
Clinical governance
Clinical governance is essential in supporting safe, high-quality health products and services and a nationally connected health and care system.
Clinical governance in digital health
Clinical governance is the combination of culture, systems, and processes that enable everyone working in digital health to support the delivery of safe, high-quality care for consumers.
Why it matters
Digital health products and services are increasing in prominence. As their use expands, so does the need for strong clinical governance practice to support how these technologies are designed, implemented and used. Clinical governance supports digital health solutions to function as intended, reducing the risk of harm, building trust and supporting positive experiences and outcomes across Australia's health and care system.
Having clinical governance systems in place can help mitigate risks and reduce their likelihood. Clinical governance also helps ensure that if something does go wrong, there are clear systems and processes in place to allow a swift and effective response. Strong clinical governance therefore ensures there is a continuous process of safety and quality improvement.
Clinical governance framework
The Clinical Governance Framework for Digital Health applies to digital health products and services. It is underpinned by 5 principles, each of which are equally important. The framework can serve as a guide for other organisations looking to develop their own clinical governance framework.
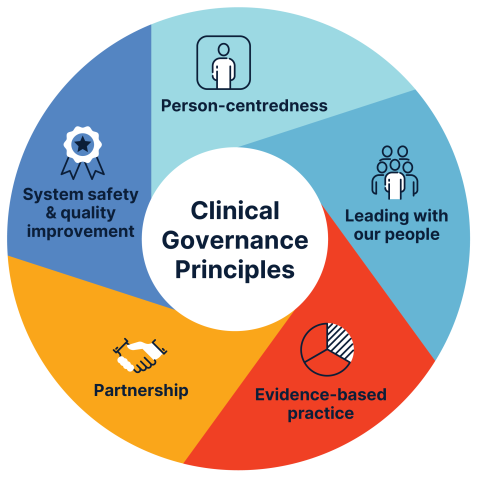
Person-centredness
Consumers' needs, goals, values, preferences and experiences inform how digital health products and services are designed, developed and delivered, supporting safe, high-quality products and services.
Leading with our people
Promoting a culture of safety, quality and continuous improvement through strong leadership at all levels of the organisation. This includes clear accountability, workforce engagement, and supporting staff to maintain the appropriate skills and qualifications for their roles.
Evidence based practice
Using evidence including internal and external data, insights and research to inform the design, development and delivery of products and services. This includes analysing consumer feedback and complaints and evaluating clinical safety incidents.
Partnership
Collaborating with key stakeholders, including people with lived experience, to design, develop and deliver products and services that reflect their needs and expectations. This means actively seeking to understand the experience, context, needs, behaviours and workflows of service users.
System safety and quality improvement
Supporting a culture of safety and continuous quality improvement and learning through:
- Early and ongoing identification, analysis and mitigation of risk.
- Incidents are recognised, monitored and managed appropriately.
- Safety and quality indicators are monitored, reviewed and analysed.
Clinical governance groups
A national committee has been established to strengthen clinical oversight and enable the highest standards of safety and quality in digital health.
The National Clinical Governance Committee – Digital Health (NCGC-DH) will guide Australia's approach to emerging technologies such as virtual care and AI in healthcare with Expert Advisory Groups. These groups will include clinicians, consumers, industry, health technology experts and other relevant government agencies like the Therapeutic Goods Administration and Australian Commission on Safety and Quality in Health Care.
